 One size quality program does not fit all. Resource, cost, and many other variables. When a resource is considering a quality management program, andĬhoosing between GLP, CLIA and ISO, there are many considerations such as if the resource has legal obligations to state and federal laws, type of No particular approach is recommended, but severalĪre mentioned below to help design the appropriate QMS for the biospecimen resource. 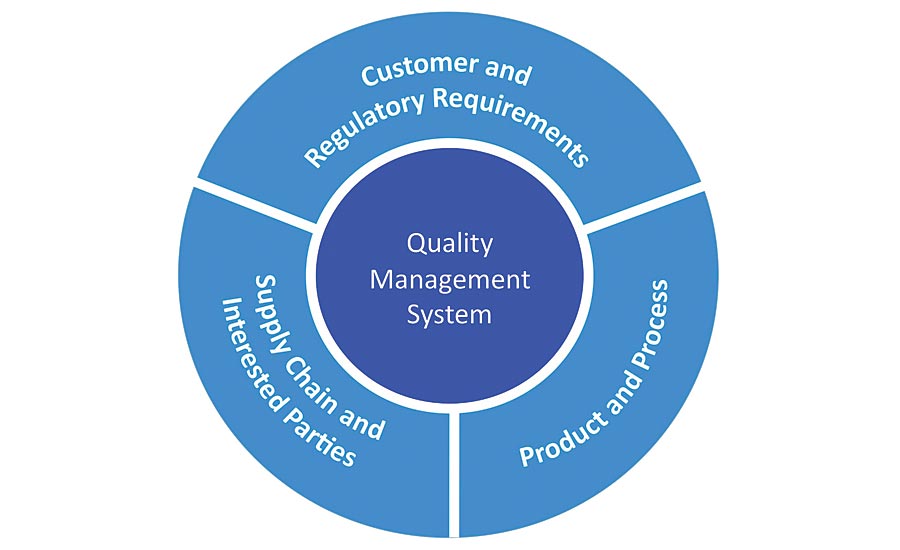
Management programs available upon which to pattern individual biospecimen resource QMS policies. Each biospecimen resource should either establishĪ written QMS or adhere to a QMS published by the organization with which the biospecimen resource is associated. The QMS describes theīiospecimen resource’s QA/QC policies and approaches for ensuring that program requirements are met. Involved in repository operations, although this might not be possible for smaller or less established biospecimen resources. 
Where feasible, the QMS should be managed by individuals who are not Quality Management Systemīiospecimen collection, processing, management, and distribution should be carried out within a quality management system (QMS) that containsĭocumented quality assurance/quality control (QA/QC) policies and written SOPs. Last Updated: 03/29/16 Quality Management B.3.1. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
